July 1, 2021
Scientific Breakthrough in Alzheimer’s is Here

Burlingame, California, July 1, 2021
Recent clinical validation of Apollo Health’s ReCODE Protocol™ — an intervention designed to reverse cognitive decline — could finally bring relief to millions who suffer from dementia. That validation comes from early results of a clinical trial based upon the science of Dale Bredesen, M.D. and recently shared in a pre-print entitled Precision Medicine Approach to Alzheimer’s Disease: Successful Proof-of-Concept Trial, posted in medRxiv, the Yale-backed pre-print server for health sciences. The trial utilized the ReCODE Protocol™ and showed the first ever improvement in cognition in early-stage Alzheimer’s patients.
In the team’s study, 84% of subjects experienced significant improvement over multiple metrics of cognition including MoCA, CNS Vital Signs, Brain HQ, AQ-21, and AQ-C. What makes ReCODE successful? Instead of pre-determining a single- pharmaceutical treatment, ReCODE identifies and targets root cause contributors to cognitive decline for each person. These assessed potential contributors include inflammation, insulin resistance, nutrient and hormonal deficiencies, specific pathogens, toxicants, and biotoxins, as well as genetics. This process has led to unprecedented results. In addition to 84% of patients experiencing a cognitive improvement, there were reported improvements in symptoms and MRI gray matter volumes as well.
While most scientific breakthroughs take decades to hit the market, ReCODE is poised to help people quickly, as it is already available for consumers via Apollo Health. There are trained practitioners in almost every state and in many international locations.
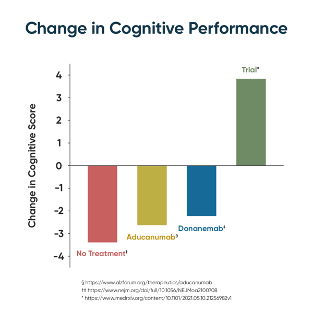
After hundreds of failed Alzheimer’s trials, and a few pharmaceutical options (such as donanemab and aducanumab) that merely slowed decline, the ReCODE protocol represents a giant leap forward in treatment and may finally bring relief to those living with dementia.
Media Inquiries:
Laura Lazzarini
media@ahnphealth.com




